You can find the atomic number from an isotope symbol.For example, if you are told the element name is aluminum, you can find the name or symbol Al to determine the atomic number is 13. While other numbers may be decimal values, the atomic number is always a simple positive whole number. There may be many numbers on a periodic table, so how do you know which one to pick? The atomic numbers go in order on the table. If you have an element name or symbol, use a periodic table to find the atomic number.Properties of sodium atoms Sodium atomic number 11 Symbol Na State at 20☌ Solid Atomic weight 22.98976928 Group 1 Period 3 Block s-block Electrons per shell 2, 8, 1 Electron configuration 1s 2 2s 2 2p 6 3s 1 Oxidation states +1 Valency 1 Valence electrons 1 Melting point 370.944 K (97.794 ☌, 208.029 ☏) Boiling point 1156.090 K (882.940 ☌, 1621.292 ☏) Atomic radius 186 pm Covalent radius 166☙ pm Van der Waals radius 227 pm Electronegativity Pauling scale: 0.93 Electron Affinity 0. Therefore, the number of neutrons in sodium(Na) is 12.īased on the atomic number of the element, the mass number, and the number of neutrons, three things can be considered. We know that the atomic number (Z) of sodium is 11 and the atomic mass number (A) is about 23. That is, neutron number (n) = atomic mass number (A) – atomic number (Z) Thus, the number of neutrons in an element is obtained from the difference between the number of atomic masses and the number of atoms. That is, the number of atomic masses (A) = p + n Therefore, the total number of protons and neutrons is called the atomic mass number. Therefore, atomic mass refers to the total mass of protons and neutrons.Ītomic mass (A) = nucleus mass = total mass of protons and neutrons (p + n)Īgain, the mass of each proton and neutron is about 1amu. The nucleus is made up of protons and neutrons. Therefore, the mass of the nucleus is called atomic mass. 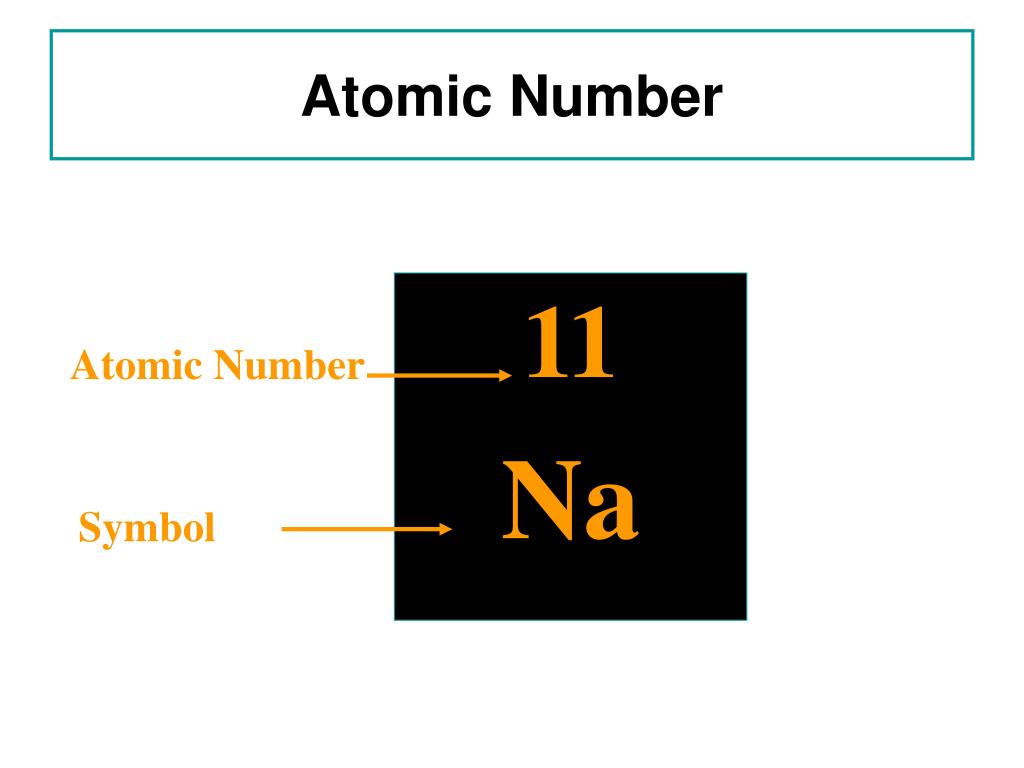
One is a positively charged particle proton and the other is a charge-neutral particle neutron.Īlmost all the mass of the atom is accumulated in the nucleus. There are two types of particles in the nucleus. We already know that the nucleus is at the center of the atom. Relationship between the atomic mass and sodium atomic number This site has published an article detailing the electron configuration of sodium that you can read if you want. To know these properties of sodium one must know the atomic number of sodium. The electron in the last shell of sodium easily forms a bond by donating to another element. Therefore, sodium is the s-block element. The last electron of sodium enters the s-orbital. So, the valence electrons of sodium are one. And the last shell has a total of an electron. The last shell of sodium has an unpaired electron, so the valency of sodium is 1. Therefore, the number of negatively charged electrons orbiting in its orbit is equal to the number of positively charged protons in the nucleus.Ītomic number (Z) = Number of charges in the nucleus (p) Importance of the atomic number of sodium That is the total number of protons in the atomic number. We know that protons are located in the nucleus of an atom as a positive charge. This number is equal to the serial number of the periodic table. The atomic number of the element is expressed by ‘Z’. Thus, the number of positive charges present in the nucleus of an element is called the atomic number of that element. He named that number the order of the atoms. The results of the experiment show that each element has a unique integer equal to the number of positive charges in the nucleus of that element. Scientist Henry Gwyn Jeffreys Moseley researched the X-ray spectrum of various elements in 1913-1914. Properties of sodium atoms What is the atomic number?.Relationship between the atomic mass and sodium atomic number.Importance of the atomic number of sodium.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
